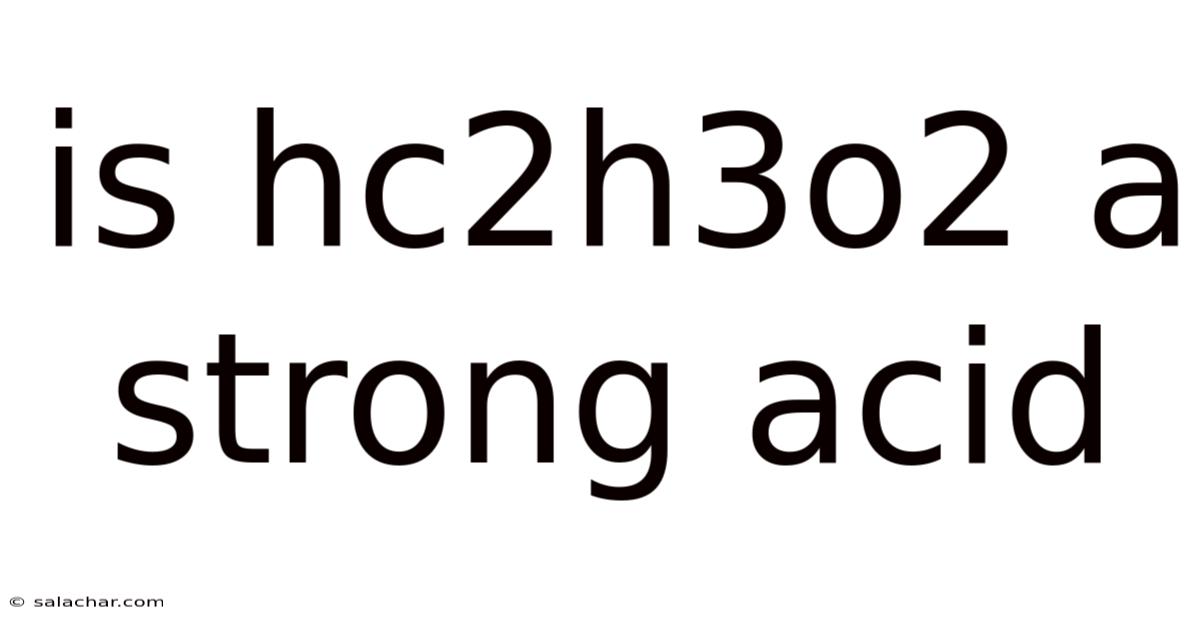Is HC₂H₃O₂ a Strong Acid? Understanding Acetic Acid and Acid Strength
Is HC₂H₃O₂ a strong acid? The short answer is no. But hC₂H₃O₂, more commonly known as acetic acid, is a weak acid. This article will delve deep into understanding why, exploring the concepts of acid strength, the properties of acetic acid, and the implications of its weak acidity. We'll also examine the chemical behavior of acetic acid and compare it to strong acids to highlight the key differences. By the end, you'll have a comprehensive understanding of acetic acid and its classification as a weak acid Most people skip this — try not to..
Introduction to Acid Strength
Before we look at the specifics of acetic acid, let's establish a foundation in understanding acid strength. Acids are substances that donate protons (H⁺ ions) in aqueous solutions. Now, the strength of an acid is determined by its ability to donate these protons. On the flip side, Strong acids completely dissociate (ionize) in water, meaning they release all their protons. In contrast, weak acids only partially dissociate, meaning they only release a small fraction of their protons. This difference significantly impacts their chemical behavior and properties.
Understanding the Dissociation of Acetic Acid
Acetic acid, the main component of vinegar, is a weak organic acid. Its chemical formula is HC₂H₃O₂. When acetic acid is dissolved in water, it undergoes a reversible dissociation reaction:
HC₂H₃O₂(aq) ⇌ H⁺(aq) + C₂H₃O₂⁻(aq)
The double arrow (⇌) indicates that the reaction proceeds in both directions simultaneously. A significant portion of the acetic acid molecules remain undissociated (HC₂H₃O₂), while only a small percentage dissociate into hydrogen ions (H⁺) and acetate ions (C₂H₃O₂⁻). This partial dissociation is the defining characteristic of a weak acid.
The Role of the Equilibrium Constant (Ka)
The extent of dissociation of a weak acid is quantified by its acid dissociation constant, K<sub>a</sub>. Day to day, K<sub>a</sub> represents the equilibrium constant for the dissociation reaction. A smaller K<sub>a</sub> value indicates a weaker acid. For acetic acid, the K<sub>a</sub> value is approximately 1.Still, 8 x 10⁻⁵. This very small value clearly demonstrates that only a tiny fraction of acetic acid molecules dissociate in water, solidifying its classification as a weak acid But it adds up..
Comparing Acetic Acid to Strong Acids
To further illustrate the difference, let's compare acetic acid to a strong acid like hydrochloric acid (HCl). Hydrochloric acid completely dissociates in water:
HCl(aq) → H⁺(aq) + Cl⁻(aq)
Notice the single arrow (→), indicating a complete and irreversible reaction. All HCl molecules dissociate into hydrogen ions and chloride ions. This complete dissociation leads to a much higher concentration of H⁺ ions in solution compared to a solution of acetic acid of the same concentration. The K<sub>a</sub> value for strong acids is very large, essentially meaning the reaction proceeds almost entirely to completion.
Properties of Acetic Acid Highlighting its Weak Acidity
Several properties of acetic acid directly reflect its weak acidity:
-
Low pH but not extremely low: While acetic acid solutions are acidic (pH < 7), their pH values are relatively higher than those of strong acids at the same concentration. This is because the lower concentration of H⁺ ions in solution results from the incomplete dissociation.
-
Weak conductivity: Acetic acid solutions are poor conductors of electricity. This is because the low concentration of ions (H⁺ and C₂H₃O₂⁻) in solution limits their ability to carry an electric current. Strong acids, on the other hand, are excellent conductors due to their high ion concentration And it works..
-
Reactions with bases: Acetic acid reacts with bases to form salts and water. Even so, the reaction is less vigorous compared to the reactions of strong acids with bases. This is because the limited availability of H⁺ ions in acetic acid solutions slows the reaction rate Which is the point..
Chemical Behavior and Applications of Acetic Acid
Despite being a weak acid, acetic acid exhibits a wide range of chemical behaviors and applications, including:
-
Vinegar production: Acetic acid is the primary component of vinegar, a common household item used for cooking and cleaning. The weak acidity of vinegar allows it to be safely used in food preparation The details matter here. Which is the point..
-
Industrial applications: Acetic acid is widely used in the production of various chemicals, including plastics, textiles, and pharmaceuticals. Its weak acidity allows for controlled reactions in many industrial processes Still holds up..
-
Buffer solutions: Acetic acid, along with its conjugate base, acetate (C₂H₃O₂⁻), can be used to prepare buffer solutions. Buffer solutions resist changes in pH when small amounts of acid or base are added. This property is crucial in many biological and chemical applications Simple, but easy to overlook..
-
Food preservation: The weak acidity of acetic acid inhibits the growth of bacteria and other microorganisms, contributing to its use as a food preservative.
Understanding the Conjugate Base: Acetate Ion (C₂H₃O₂⁻)
When acetic acid dissociates, it forms the acetate ion (C₂H₃O₂⁻). The acetate ion can react with H⁺ ions to reform acetic acid, showing the reversible nature of the dissociation reaction. Understanding the properties of the conjugate base helps explain the behavior of acetic acid in solution. This ion is the conjugate base of acetic acid. The stability of the acetate ion also contributes to the relatively weak acidity of acetic acid And it works..
Frequently Asked Questions (FAQ)
Q: What makes an acid strong or weak?
A: The strength of an acid is determined by its extent of dissociation in water. Which means strong acids completely dissociate, while weak acids only partially dissociate. This is reflected in their K<sub>a</sub> values: Strong acids have very large K<sub>a</sub> values, while weak acids have small K<sub>a</sub> values And it works..
Q: Is acetic acid dangerous?
A: Concentrated acetic acid (glacial acetic acid) can be corrosive and cause skin burns. Even so, dilute solutions of acetic acid, like vinegar, are generally safe for household use But it adds up..
Q: Can acetic acid be neutralized?
A: Yes, acetic acid can be neutralized by reacting it with a base. This reaction produces a salt and water. Here's one way to look at it: reacting acetic acid with sodium hydroxide (NaOH) produces sodium acetate (NaC₂H₃O₂) and water Still holds up..
Q: What is the difference between pH and pKa?
A: pH measures the acidity or basicity of a solution, representing the concentration of H⁺ ions. On top of that, pKa, on the other hand, is a measure of the acidity of a specific acid, reflecting its tendency to donate protons. Plus, the pKa value is the negative logarithm of the K<sub>a</sub> value. A lower pKa indicates a stronger acid Nothing fancy..
Q: How does the structure of acetic acid contribute to its weak acidity?
A: The structure of acetic acid plays a significant role in its weak acidity. That said, the methyl group (-CH₃) attached to the carboxyl group donates electron density to the carboxyl group, reducing the ease of proton donation and thus weakening the acidity. The presence of the carboxyl group (-COOH) is responsible for the acidic nature. This electron donation stabilizes the acetate ion, which also contributes to the weaker acidity.
Conclusion: Acetic Acid – A Weak but Important Acid
At the end of the day, HC₂H₃O₂, or acetic acid, is definitively a weak acid. Its incomplete dissociation in water, as evidenced by its small K<sub>a</sub> value (1.8 x 10⁻⁵), low conductivity, and relatively high pH compared to strong acids at the same concentration, clearly establishes its classification. On the flip side, its weak acidity doesn't diminish its importance. Still, acetic acid matters a lot in various industrial processes, food production, and even in biological systems. Understanding its properties and behavior is essential in diverse fields, from chemistry and biology to food science and industrial manufacturing. Its unique combination of weak acidity and versatility makes it a substance worthy of detailed study and appreciation Surprisingly effective..